Epidemiological Profile of COVID-19 Cases and Vaccination in the Federal Public Prosecutor’s Office (MPF) in November 2021
Presentation
Through this study, entitled “COVID-19 Research in the Federal Public Prosecutor’s Office (MPF)”, the Department of Integrated Health Services (SSI-Saúde) sought to map the epidemiological profile of COVID-19 cases within the Federal Public Prosecutor’s Office (MPF), as well as to gather information regarding vaccination. The results will support decision-making, enhancing health promotion actions for the internal audience and, indirectly, for the general population.
The research was conducted at the national level between October 25 and November 5, 2021, with dissemination carried out by the Institutional Communication Department (Secom/PGR). The target audience consisted of MPF members, employees, and interns.
The study covered data on participants’ profiles (gender, age, and assigned unit), confirmed cases of the disease, severity of cases, aftereffects, diagnostic methods, as well as vaccination data. Participation was not mandatory, and no data capable of identifying participants were collected.
For data collection, the LimeSurvey tool was used. Data and texts were processed using the Python programming language. The charts presented in this document were generated using Jupyter Notebook (through the Matplotlib and Seaborn libraries) and Microsoft Power BI. The analysis and knowledge generation were carried out using programming, statistics, data mining, natural language processing, plotting, and algorithm application.
This work’s integration effort involved participation from all MPF units, regardless of whether they had an available health service. Units were encouraged to take part in the research, ensuring a representative number of responses and, consequently, an analysis that accurately reflects the Institution as a whole.
The information presented in this report was consolidated by SSI-Saúde, the department responsible for planning and executing actions related to the pandemic. The results hold national statistical representativeness, as well as specific representativeness for the Federal District and the states of São Paulo and Rio de Janeiro, with a 5% margin of error.
Objectives
Applied across all units, the research aimed to understand the impact of COVID-19 on the Federal Public Prosecutor’s Office (MPF), as well as the vaccination status, according to the objectives listed below:
- Identify the epidemiological profile of COVID-19 cases: Determine the gender, age group, and region most affected by the disease, in addition to assessing the severity of health conditions among the surveyed target audience.
- Analyze the aftereffects of COVID-19: Map the main post-COVID-19 sequelae among internal personnel and evaluate their characteristics according to the epidemiological profile within the Institution.
- Assess the vaccination coverage profile: Identify the vaccinated population’s characteristics, correlating the types of vaccines administered and the most frequent side effects.
Sample Profile
All units participated in the COVID-19 Research in the Federal Public Prosecutor’s Office (MPF), totaling 3,026 responses. However, only complete responses were considered, amounting to 2,866 (two thousand eight hundred and sixty-six) valid responses, representing 95% of the received answers. This sample size is statistically representative of the MPF personnel population.
In this report, only male and female genders were considered, given the low representativity of other gender classifications (less than 0.1%). Additionally, the study aims to correlate chromosomal aspects with COVID-19 cases.
Regarding the age of participants, the average age is 49 years, with the majority being employees (91%).


Composed of interns, members, and employees, the survey’s target audience is evenly distributed by gender. More than 70% of participants are between 36 and 56 years old (Figure 3).

The survey participants are between 20 and 74 years old. To define age groups, they were divided into decades, with the highest concentration in the 40 to 49-year-old range.

The epidemiological profile of this study was structured based on responses from MPF units across all regions of Brazil.
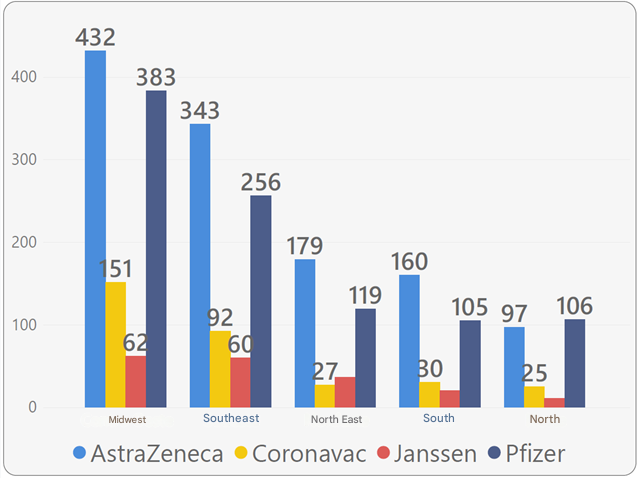
Figures 5 and 6 below present the distribution of survey participants, considering the Brazilian regions (Central-West, Southeast, Northeast, South, and North) in descending order based on the number of participants.


Epidemiological Profile
This section provides details on the epidemiological profile of participants who had a confirmed COVID-19 diagnosis.
Confirmed cases account for 24% of the sample, a figure that is nationally representative for the MPF, as well as for the units in Rio de Janeiro, São Paulo, and the Federal District.

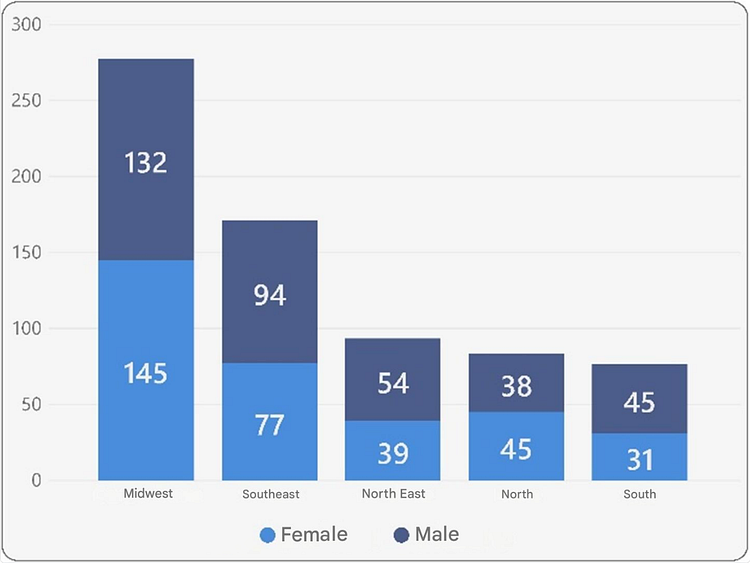
The frequencies by gender, position, region, age group, and age are proportionally distributed between the total population of participants and the confirmed cases, indicating no correlation between these variables and the disease.


To perform the correlations in this study, the Pearson correlation coefficient was used due to the linear nature of the analyzed variables.
The correlation between confirmed COVID-19 cases and gender or age group is nonexistent (p < 0.05).

The proportional distribution of gender, age groups, and ages in the MPF population and confirmed COVID-19 cases indicates that the disease affected the target audience equally, regardless of these variables.
However, to extract further insights from the confirmed cases, a clustering analysis was performed using the K-Means Machine Learning algorithm. This analysis confirmed the proportionality of COVID-19 cases according to the studied variables.
Additionally, it revealed that severe COVID-19 cases (requiring hospitalization), although proportionally distributed across the country’s regions, were more prevalent among men in the 50-year-old age group.
Disease Severity
The study analyzed the severity of the disease and the presence of aftereffects among participants who reported confirmed COVID-19 cases (24% of participants).
The severity of the disease was measured in three levels:
- (I) Asymptomatic (“Not applicable”) — Cases where there was either no confirmed diagnosis or a confirmed diagnosis without symptoms.
- (II) Mild cases — Cases without hospitalization.
- (III) Severe cases — Cases requiring hospitalization due to COVID-19.
The majority of participants and confirmed cases presented mild symptoms. The distribution of severity levels can be observed in the figures below.



Although there is no correlation between gender or age group and the number of COVID-19 cases, a higher number of severe cases (hospitalizations) was observed among men, particularly in the 50 to 59-year-old age group.
Meanwhile, mild cases were more concentrated in the 40 to 49-year-old range, for both men and women.
Aftereffects
Among the 24% of participants who tested positive for COVID-19, 37% reported experiencing at least one type of aftereffect.
The reported aftereffects were categorized and ranked in descending order according to their frequency.
Among the surveyed population, the most common aftereffects were: memory loss, anosmia (loss of smell), asthenia (fatigue), hair loss, and ageusia (loss of taste) (Figure 15).
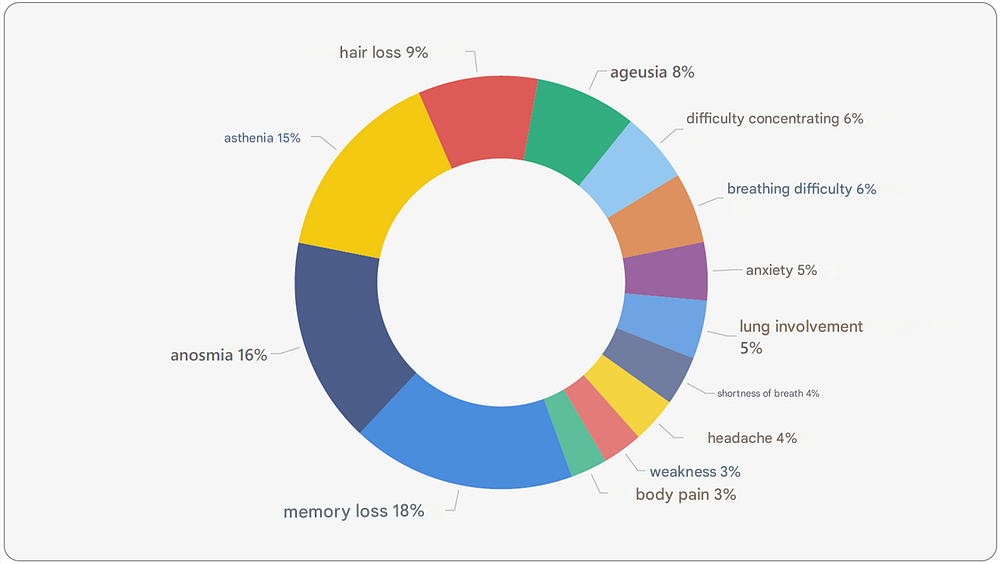
Many aftereffects could have been grouped together, such as “respiratory difficulty” and “shortness of breath”.
However, in this case, they were kept separate because the former is slightly broader than the latter, considering the possibility of other respiratory difficulties that are not limited to shortness of breath.
Some aftereffects were standardized by replacing terms as follows:
- “Loss of smell” was replaced with “anosmia”.
- “Excessive tiredness” was replaced with “asthenia”.
- “Loss of taste” was replaced with “ageusia”.
- “Difficulty with attention/reading” was replaced with “difficulty concentrating”.
- “Excessive and insatiable thirst” was replaced with “hyperhidrosis”.
In addition to the most frequent COVID-19 aftereffects indicated in Figure 15, the complete list of aftereffects reported by surveyed participants who confirmed a COVID-19 diagnosis is provided below, arranged in alphabetical order.
Reported Aftereffects
Acid reflux, ageusia, allergy, amenorrhea, anosmia, anxiety, arm pain, asthenia, asthma, back pain, blood clotting issues, blood sugar issues, body pain, boils, brittle nails, bursitis, cardiac arrhythmia, cataract, chest pain, cholera, circulatory problems, cognitive deficit, cough, decreased libido, depression, dermatological disorder, diabetes, difficulty, difficulty concentrating, digestive issues, distress, dizziness, elbow pain, emotional disorders, fear, fever, gastrointestinal disorder, hair loss, hallucinations, hand tremors, headache, high blood pressure, high cholesterol, hyperhidrosis, hypersomnia, hypertension, insomnia, intestinal problems, irritability, joint pain, kidney problems, knee pain, labyrinthitis, leg pain, leg thrombosis, liver problems, loss of appetite, loss of balance, low immunity, low oxygen saturation, lung impairment, memory loss, menstrual irregularity, mental confusion, migraine, muscle mass loss, muscle pain, muscle stiffness, myasthenia, myocarditis, nausea, nausea, nervousness, neurasthenia, neurological problems, neuropathy, palmar hyperhidrosis, panic attacks, panic disorder, plantar hyperhidrosis, pneumonia, polydipsia, psychological disorders, pulmonary secretion, reasoning difficulty, respiratory difficulty, rhinitis, rib pain, rough tongue, shortness of breath, shoulder pain, sinusitis, sleep apnea, sleep disorder, snoring, spinal problems, stress, tachycardia, temperature fluctuation, tendinitis, thigh sensitivity, thyroid disorder, tingling sensation, tinnitus, urinary tract infection, varicose veins, vertigo, vision problems, vomiting, weakness, weight loss.
Testing
The COVID-19 Research in the Federal Public Prosecutor’s Office (MPF) also analyzed the methods used to detect the disease.
It was found that 49% of participants reported having undergone some form of testing to confirm a COVID-19 diagnosis.
The majority of tests were performed using the nasal swab (PCR) method, as shown in the figures below.

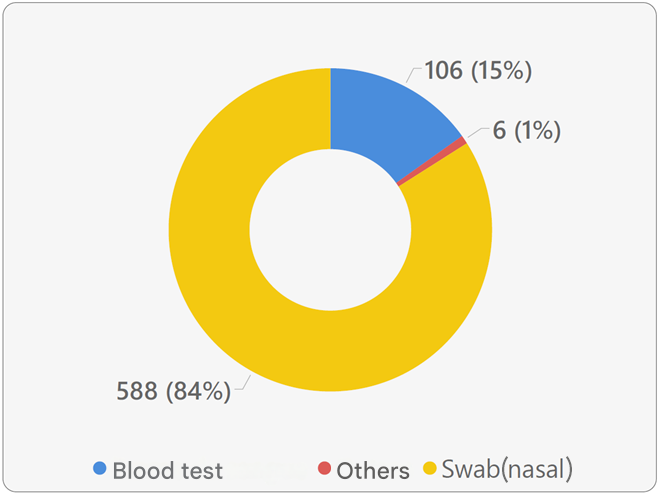
Two different methods were used to confirm the COVID-19 diagnosis among the survey participants, namely blood tests and nasal swab (PCR) tests.
Among the confirmed COVID-19 cases (24% of participants) — excluding those who did not undergo any testing — the proportion of testing methods remained consistent:
- 84% underwent nasal swab (PCR) testing
- 16% underwent blood testing
These proportions align with the overall testing distribution among all participants (42% nasal swab and 7% blood test).
Vaccination
As of November 5, 2021 (the last day for submitting the survey form), approximately 97% of participants had received at least one dose of a SARS-CoV-2 vaccine.
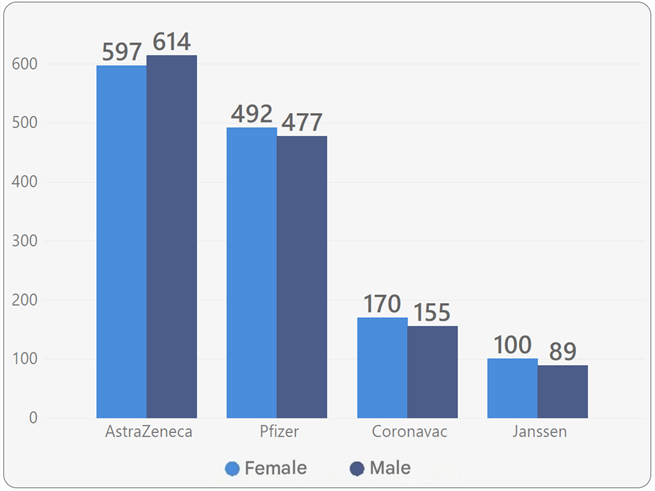
The majority of vaccinated participants received AstraZeneca and Pfizer vaccines.

A total of 3% of participants had not received any vaccine.
Another 3% had received a combination of two different vaccine brands.
Meanwhile, 94% of participants had received one or two doses of the same brand (AstraZeneca, Pfizer, CoronaVac, or Janssen).

Among the confirmed COVID-19 cases, the AstraZeneca and Pfizer vaccines were also the most commonly administered, accounting for 75% of cases.
The vaccination coverage among the MPF population is proportionally distributed according to gender, position, age group, and the geographic region of the participants’ assigned unit.


Participants who received at least one dose of a vaccine reported the most common adverse reactions they experienced after vaccination.
Among the listed reactions, the five most frequent were:
- Chills
- Malaise
- Itching
- Nasal congestion
- Diarrhea
The frequency of adverse reactions can be observed in Figure 24.

In order to deepen the understanding of the adverse effects of the vaccines administered to the MPF population, these effects were analyzed individually by vaccine type, as described below.
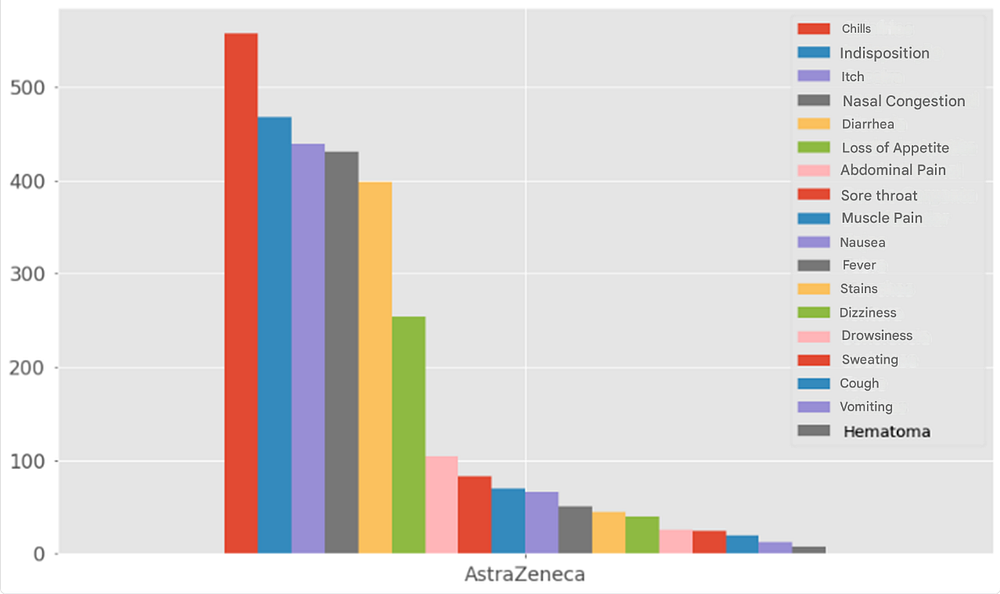
AstraZeneca
The AstraZeneca vaccine was the most widely administered (41% of participants received it).
The main reported reactions were:
- Chills (18%)
- Malaise (15%)
- Itching (14%)
- Nasal congestion (14%)
- Diarrhea (13%)
- Loss of appetite (8%)
Pfizer
The Pfizer vaccine was the second most administered, with 34% of participants receiving it.
The main reported reactions were:
- Itching (27%)
- Chills (19%)
- Nasal congestion (14%)
- Loss of appetite (10%)
- Malaise (8%)
- Diarrhea (7%)

CoronaVac
The CoronaVac vaccine was the third most administered, with 12% of participants receiving it.
The main reported reactions were:
- Itching (25%)
- Chills (20%)
- Loss of appetite (11%)
- Nasal congestion (10%)
- Malaise (6%)
- Abdominal pain (5%)

Janssen
The Janssen vaccine ranked fourth and was the least administered, with only 7% of participants receiving it.
The main reported reactions were:
- Chills (19%)
- Diarrhea (17%)
- Malaise (15%)
- Nasal congestion (12%)
- Itching (11%)
- Loss of appetite (10%)

It was also found that the majority of participants (83%) had already received two doses of a vaccine.
Single-dose vaccinations correspond to the Janssen vaccine (7%).
While younger participants received one dose, a single-dose vaccine, or two doses, older participants received two doses or two doses plus a booster (Figures 29 and 30).


The 3% of MPF members who did not receive a vaccine were categorized into five groups:
- Opposition — Those who are ideologically opposed to vaccination.
- Waiting — Individuals who are waiting for the testing phase to be completed.
- Already had the disease — Those who believe that because they have already contracted the virus, they possess antibodies and therefore do not need the vaccine.
- Adverse effects — Those who are concerned about potential side effects of the vaccines.
- Medical recommendation — Individuals with health conditions who, based on medical advice, should not receive the vaccine.

Conclusions
Conducted by SSI-Saúde, the COVID-19 Research in the Federal Public Prosecutor’s Office (MPF) provided insight into the epidemiological profile of COVID-19 within the MPF, including the analysis of aftereffects and vaccination.
The distribution of responses by gender, age, age group, and region ensured a sample representative of the MPF personnel population.
Based on the knowledge produced, the following conclusions can be drawn:
- 24% of MPF personnel were diagnosed with COVID-19. Most of them (89%) reported experiencing a mild form of the disease, meaning no hospitalization was required. The disease affected MPF personnel equally, regardless of gender, age, age group, or assigned region.
- The most affected group in terms of severe COVID-19 cases (requiring hospitalization) consisted of male individuals, particularly those aged 50 to 59 years.
- The most common aftereffects of COVID-19 were memory loss, anosmia (loss of smell), asthenia (excessive fatigue), hair loss, ageusia (loss of taste), and difficulty concentrating. No correlation was found between disease severity and its aftereffects.
- Around 50% of MPF personnel underwent some form of testing to confirm a COVID-19 diagnosis. The majority (84%) used the nasal swab (PCR) method.
- By the final date of the study, 97% of MPF personnel had received at least one dose of the COVID-19 vaccine. The vaccination coverage profile was proportionally distributed across gender, age group, age, and regions of the country. The most commonly administered vaccines were AstraZeneca (42%) and Pfizer (34%).
- Among the most frequently reported adverse reactions, “chills” was the most common among those who received at least one vaccine dose, followed by “malaise,” “itching,” “nasal congestion,” “diarrhea,” and “loss of appetite”.
- The reasons for not getting vaccinated, reported by 3% of participants, included: Opposition to vaccination, Waiting for the testing phases to be completed, Belief that prior infection provided sufficient immunity, Concerns about side effects, and Medical recommendation against vaccination.
- Due to the low number of responses, there was no statistical representativity for MPF members and interns. However, the data described in this report is statistically representative of MPF employees.
By Juliano Oliveira and Prof. Alan Ferreira, PhD.
Links


